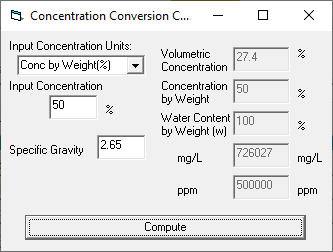
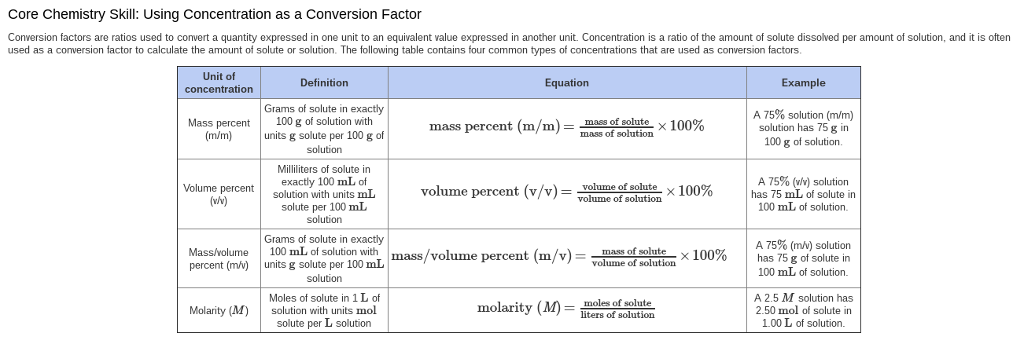
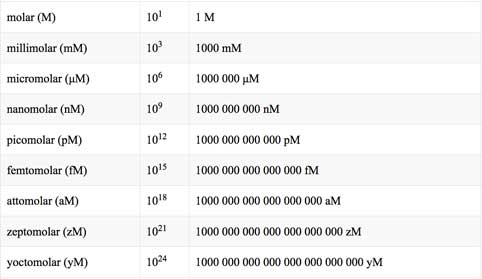
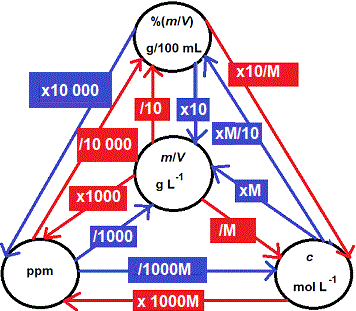
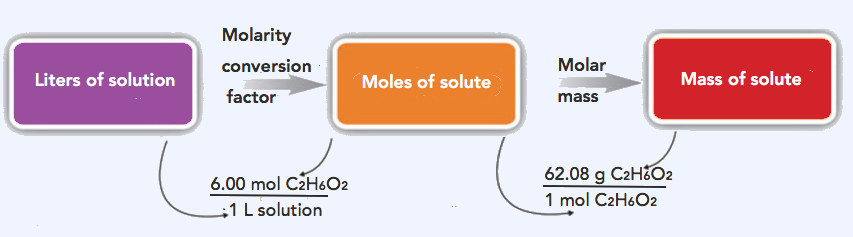
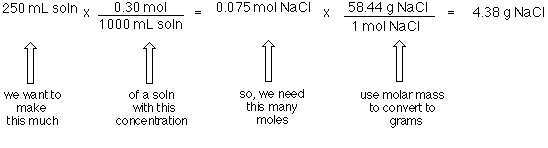SOLVED: The units for the concentration of the glucose are ug/dL, convert these units into molarity units, mol/L.
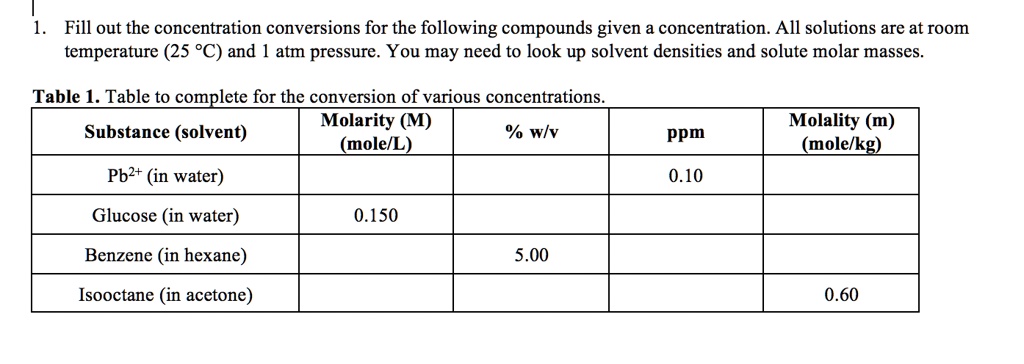
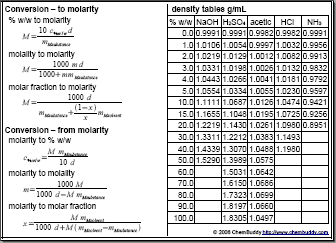
SOLVED: Fill out the concentration conversions for the following compounds given a concentration. All solutions are at room temperature (25 °C) and atm pressure. You may need to look up solvent densities