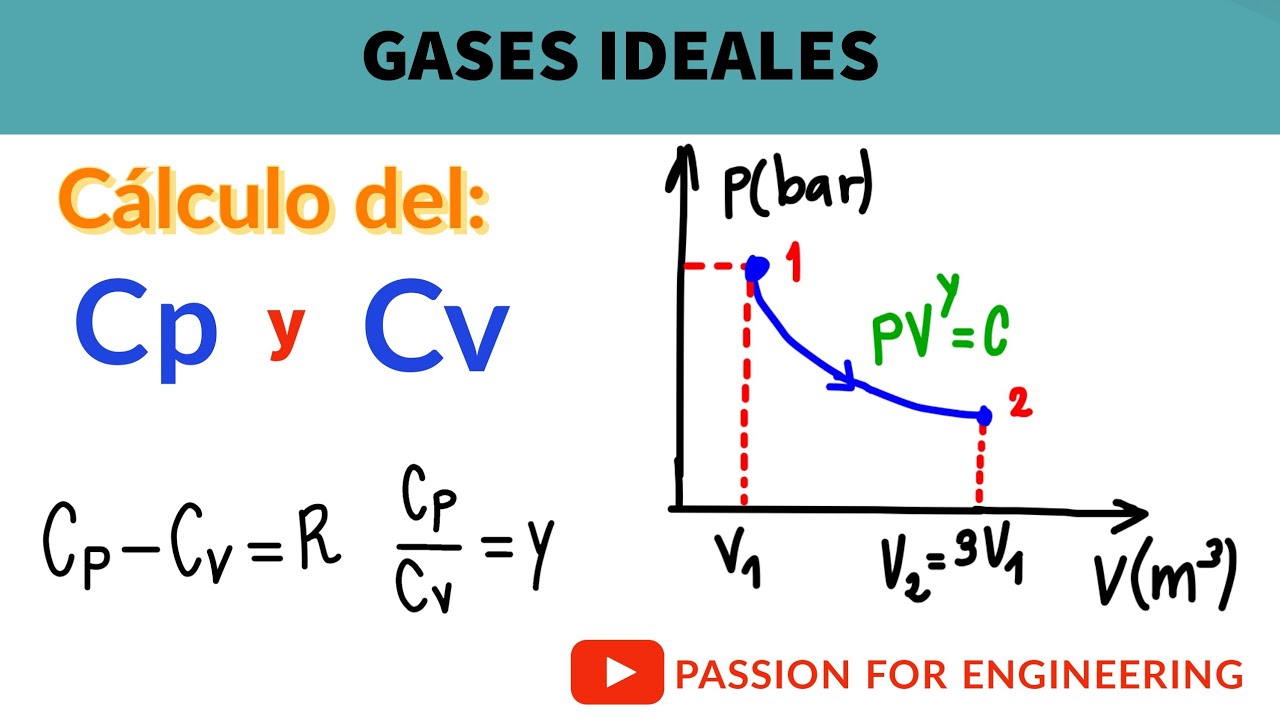
22. 1 mole of an ideal gas A (CV=3R) and 2 mole of an ideal gas B are (CV=3/2R) taken in a container and expanded reversible and adiabatically from 1 litre to
Unit Four – First Law for Ideal Gases Outline Unit Four Goals Unit Four Goals Continued Unit Four Goals Continued Why Use Idea

Fisicoquímica 1-primer principio y los gases ideales - CLASES DE FARMACIA EN SALAMANCA/profesor Pepe Blasco

Calculate the value of gamma = Cp / Cv for a gaseous mixture consisting of v1 = 2.0 moles of oxygen and v2 = 3.0 moles of carbon dioxide. The gases are assumed to be ideal.