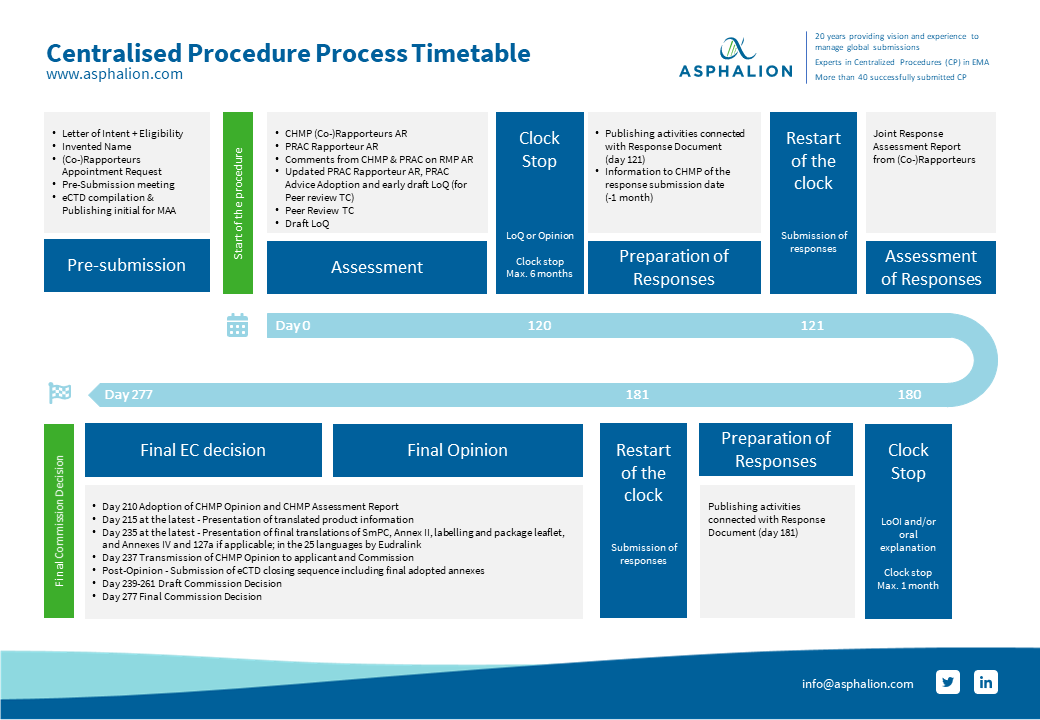
How to design a regulatory strategy to optimize registration of Advanced Therapy Medicinal Products (ATMPs) with European Medicines Agency ? - BlueReg Group

Impact of the European Union on access to medicines in low- and middle-income countries: A scoping review - The Lancet Regional Health – Europe